The role of calcification in neutralizing acidification
The biological formation of carbonate minerals plays an important role in ocean acidification recovery, a new Nature Geoscience article by NESSC-researcher Jack Middelburg and colleagues posits. Changes to the different calcification processes significantly alter the oceanic response to acidification caused by enhanced CO2 levels. A better understanding of this dynamic, dubbed biological compensation, is essential to better predict the evolution of the oceans in response to the current increase of atmospheric CO2.
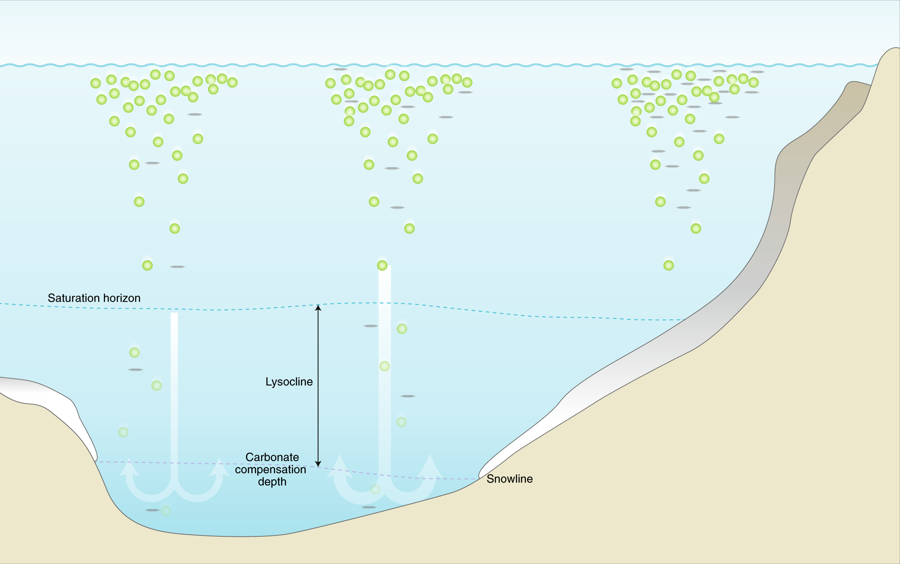
Carbonate minerals, stored in the world’s oceans as sediments or suspended in the water column, play an important part in neutralizing the acidification of ocean water under the influence of enhanced CO2-levels. The majority of these carbonate minerals is formed through biological processes, by organisms such as foraminifera, molluscs or corrals. However, accumulated evidence is showing that the production of these calcifying processes is impacted by changes in the chemistry of the surrounding waters or in response to global warming.
Different recovery scenarios
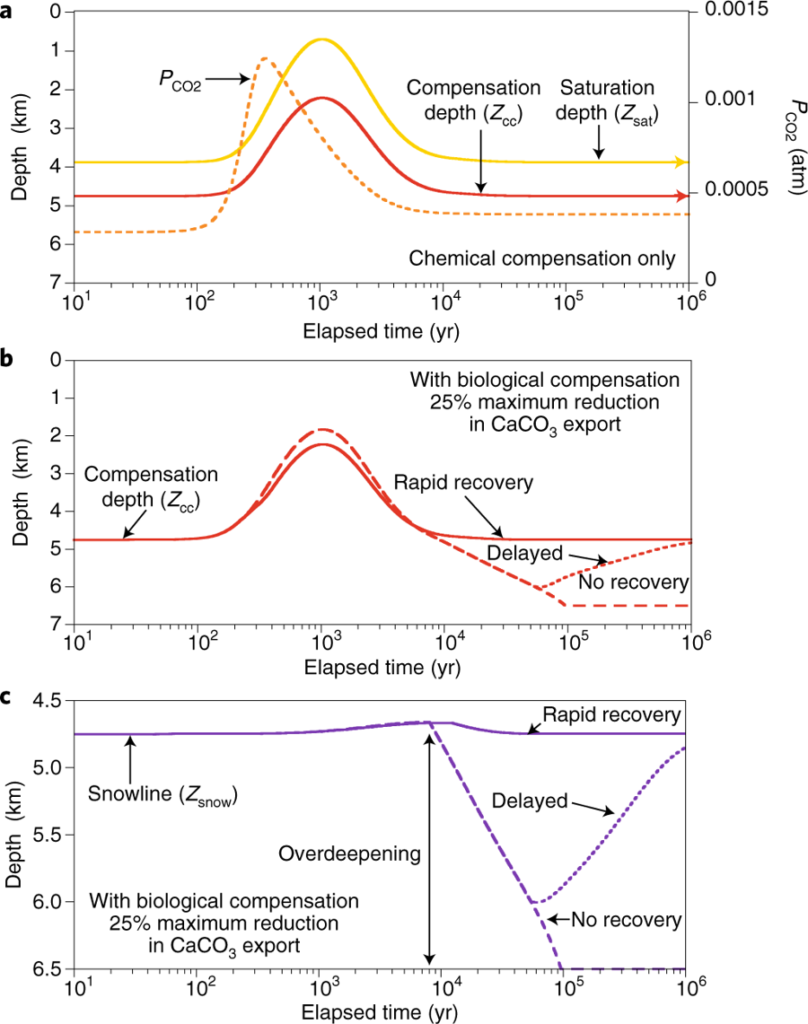
The new Nature Geoscience-publication considers the mechanisms of biological production of carbonate minerals in response to the acidification of ocean waters. The authors show that even modest drops in calcification can lead to very different recovery scenarios, from a rapid recovery, wherein calcification processes are able to keep up with increasing CO2-emissions, or to a scenario of no recovery, where calcification production falls and does not increase again.
The article concludes that a far better understanding of the processes influences biological compensation is needed to credibly predict the future carbonate chemistry of the oceans. The article further underlines that more efforts are needed to be properly incorporate calcification dynamics into long-term predictive models.
Article:
The role of calcification in carbonate compensation
P. Boudreau, J. J. Middelburg & Y. Luo
Nature Geoscience, 11, 894-900 (2018)