Climate change risks runaway algal blooms
Enhanced recycling of phosphate nutrients can starve large parts of the oceans of oxygen.
New NESSC-research by geoscientists of Utrecht University shows how climate change can trigger runaway algal blooms in the oceans. The researchers discovered that during intense periods of climate change almost no phosphate, a key nutrient, was buried in the sea floor. This caused a widespread growth of oceanic algae, starving the water of nearly all oxygen and creating large uninhabitable zones. In their article, published in Science Advances, the researchers underline that current climate change also risks an acceleration of algal blooms and oxygen loss in the oceans.
Millions of years ago, when concentrations of green-house gasses in the atmosphere were much higher than today, the oceans were warmer and more acidic. Large parts of the oceans contained no oxygen: an explosive growth of oceanic algae, succeeded by their decomposition on the sea floor, starved the ocean water of oxygen. These areas of the ocean, called ‘dead zones’, were largely uninhabitable. Also today, areas form in the ocean with oxygen concentrations so low that fish and most other marine life can’t survive. These dead zones have been observed all over the world, for example in the Baltic Sea or the Gulf of Mexico.

Nutrients
Nutrients in manure and fertilizers, like nitrogen and phosphate, improve plant growth on land. However, when these nutrients reach the coast, they boost large, and often harmful, algal blooms in the oceans. After the algae die and sink to the sea floor, their decomposition uses up all oxygen in the water, creating or expanding dead zones.
Climate change and warming ocean water also play a part in this. Geoscientists at Utrecht University were therefore interested to learn how climate change influences the formation of dead zones. To study this, the scientists looked at various chemical and biological indicators in seafloor sediments that formed millions of years ago, when greenhouse gas concentrations in the atmosphere were far higher than today and the oceans were much warmer.

Apatite minerals
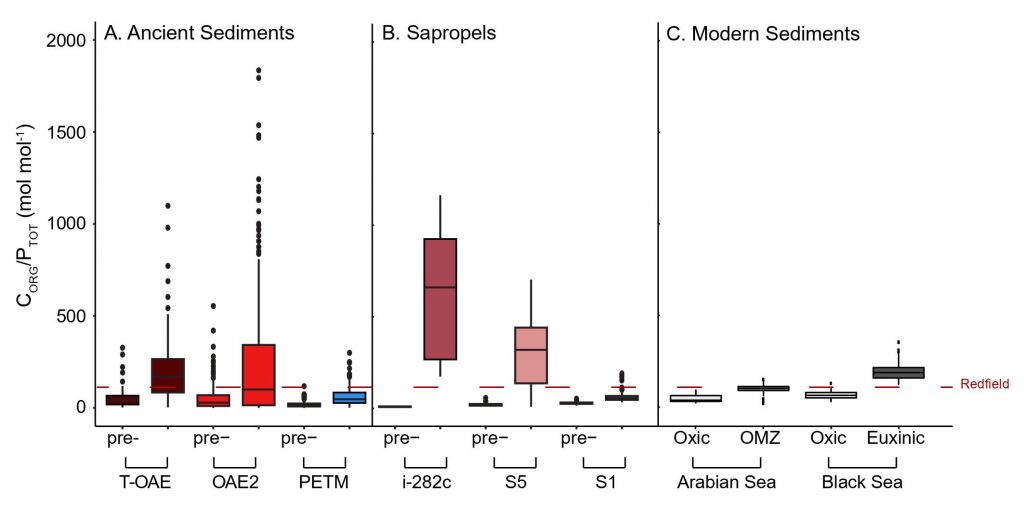
The scientists were particularly interested in learning whether the availability of phosphate for algal growth was different in these warmer conditions. When they looked at the recycling of phosphate they discovered something remarkable: in warmer oceans the nutrient phosphate was recycled much more efficiently. Less phosphate than expected was getting buried in the ocean floor. As result, phosphate kept returning to the water column where it could keep boosting algal growth.
NESSC-researcher Nina Papadomanolaki, lead author of the study, explains: “Normally, when algal remains decompose on the seafloor, the phosphate becomes stored in a mineral, apatite. Once buried in the sea floor, phosphate can no longer contribute to fueling algae growth. However, during these past periods with warmer and more acidic oceans, only very small amounts of apatite were forming in the sea floor. More phosphates molecules from decomposing algae were able to recycle back into the water again and again.”
Adverse effects
Although the scientists were not yet able to pinpoint what exactly made the forming of apatite minerals stop, their findings already have important implications for the effects of current climate change. Papadomanolaki: “This work has important ramifications for our understanding of the chemistry and biology of the past and future ocean. The increasing concentrations of CO2 in the atmosphere also risks an increasing availability of nutrients, leading to an acceleration of oxygen loss in Earth’s oceans, with all kinds of adverse effects on marine life.”
Article:
Enhanced phosphorus recycling during past oceanic anoxia amplified by low rates of apatite authigenesis
Nina M. Papadomanolaki, Wytze K. Lenstra, Mariette Wolthers and Caroline P. Slomp
Science Advances, 2022